


Changing demand behaviour (e.g. vegetarian and vegan products), sustainability, and increasing population growth rate make it necessary to develop high-quality, durable, sensory-pleasant and innovative foods.
In this context, our current research focus is on developing foods with alternative protein sources and non-alcoholic and fermented beverages in our industrial test kitchen. We focus on sustainability and the more efficient use of resources along the value chain.
Another focus of our group is on the reuse of byproducts from the food industry. The main focus is developing innovative foods and identifying technologically and nutritionally relevant ingredients of natural origin and their semisynthesis.
We also examine the changes during storing and processing (conventional and innovative techniques) on food (ageing and process markers, sensory evaluation, microbiology). The determination of authenticity markers is also part of research in the field of food quality.
We use cell culture experiments and molecular biological techniques to determine the toxicological hazards, bioavailability and beneficial effects of foods and their enriched/isolated components.
Our research aims to produce sensory-appealing foods, especially from side streams with high nutritional values, in cooperation with the industry. Another aim is to identify and isolate food ingredients that can be used as markers for ageing, processing and authenticity.
To work on our research questions, we use our expertise in food analysis, microbiology, irradiation, sensory evaluation, toxicology, bioavailability, molecular biology, cell biology, isolation and structure elucidation.
What does our research work look like? Discover it in the video:
Food development



- Development of innovative beverages, e.g. on a fermentation basis
- Development of protein-rich foods with alternative protein sources
- Development of plant-based foods (e.g. imitation cheese)
Project: CA18101 - SOURDOugh biotechnology network towards novel, healthier and sustainable food and bIoproCesseS"
Food microbiology
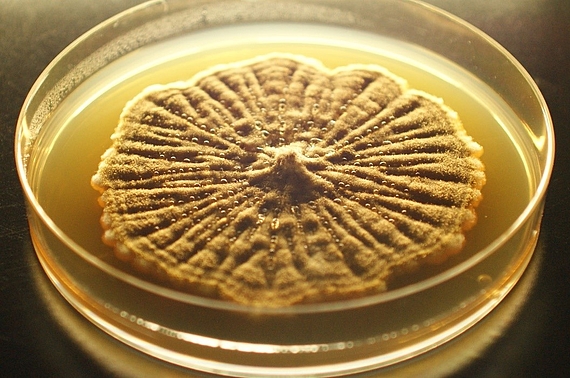
In our food microbiology laboratory, we carry out research and commissioned work with the following focuses:
- Detection of pathogenic and spoilage microorganisms in food, raw food materials, surfaces, etc.
- Load and challenge tests of food with pathogens and spoilage agents for risk assessment
- Rapid microbiological detection methods - development and validation
- Use of UV-C radiation for microbes reduction
- Use of decontamination methods (hydrogen peroxide, chlorine dioxide, UV-C radiation) for microbes reduction
- Microbiological and food hygiene consulting
- Testing and consulting on-air, personnel and environmental hygiene
Food analysis



- Extraction of food ingredients (e.g. by ultrasound, SFE)
- Qualitative and quantitative determination of food ingredients
- Method optimization, development and validation using: (U)HPLC (DAD, fluorescence), LC-MS, GC-FID, GC-MS
- activity-guided fractionation
- Semisynthesis and isolation of unknown compounds as a process, ageing or authenticity markers
- Influence of storage and processing on the spectrum of ingredients (heat, steam cooking, etc.)
- Authenticity determinations
- Structure elucidation
In vitro bioavailability and bioactivity
- Verification of toxicological safety
- Testing of in vitro bioactivity in cell culture studies, such as antioxidant, anti-inflammatory, anti-diabetic, anti-carcinogenic effects etc. at gene and protein level
- In vitro gastrointestinal digestion (determination by UHPLC)
- In vitro bioavailability in the Caco-2 cell model
- Determination of lipid peroxidation (BODIPY assay, MDA by HPLC)
- Determination of antioxidative enzymes such as SOD, CAT, GPx
Methods and equipment
An overview of the equipment of our institute, as well as various methods that we use to answer our research questions is given here.
Food development
-
Pilot plan station for food production
The equipment of the technical centre for food production is based on gastronomic facilities. Product development and food production, as well as teaching, take place here. Adjacent to the pilot plant is the rheology, sensory analysis, chemical analysis, and food microbiology facilities. The proximity enables comprehensive training in quality management, technology, research and development of new products.
The following equipment is available: household appliances (blenders, etc., refrigerators), meat grinder, cutter, gas and induction stove, tilting frying pan; combi steamer, tabletop grill, salamander, commercial microwave, deep fryer, Flexichef (multifunctional cooking device), blast chiller, climatic cabinet, Pacojet 2 Plus, BioChef Arizona Sol dehydrator, Muurikka tabletop smoking oven with thermostat, Blendtec high-performance blender, vacuum chamber machine, rheometer, viscometer, water activity meter, texture analyzer, muffle oven, Ultra-Turrax, Thermomix TM6, various juice technologies (project “Leibniz Drinks”), fermenter (up to 60 degrees)
- Sensory evaluation
-
Colorimeter
CM-5 Spektrophotometer, Konica Minolta
aw water actitivity device
Food microbiology (Bio II / S I laboratory)
-
Determination of microorganisms
In the BIO II laboratory, microbiological methods are applied and tested to identify and quantify pathogenic, spore- or toxin-producing and spoilage microorganisms. In addition to conventional methods, various rapid methods are used, applied and validated. Applications for the Greenlight™ system have been developed to detect total counts in meat, vegetables and milk products to detect Pseudomonas aeruginosa and the VBNC state in water, a dead-live detection by q-PCR and fluorescence in situ hybridization was established.
-
Reduction of microorganisms
Various UV-(C) and blue light LED-lamps
Food analysis
-
Analytical chromatography
- (U) HPLC with DAD: 190 - 640 nm and fluorescence detector: 120 - 1200 nm (Agilent 1290 Infinity LC System)
- HPLC with DAD: 190 - 950 nm, fluorescence: (Ex: 200-700 nm, Em: 280-900 nm) refractive index detector (Agilent 1200 Infinity LC System)
- HR UPLC-ESI / APCI MS: QTof Premier
-
Extraction techniques
- Ultrasound (Bandelin Sonopuls HD 2200.2, Frequence: 20 kHz, Amplitude range: 10 – 100 %, 2 – 1000 mL)
- Low pressure (0.1-1 bar) or atmospheric barrier discharge (DBD) cold plasma (plasma system type „femto“ with vacuum pump and rotary glas chamber, 13.56 MHz generator, 0-100 W; Diener electronic, Ebhausen, Germany)
-
Preparative chromatography
- Flash chromatography
- Membrane chromatography
- Countercurrent chromatography: Quattro CCC™ QuikPrep™ Chassis Mk11 with two bobbins made of stainless steel with 2.1 mm i.d. (Coil 1= 146 mL; Coil 2=148 mL) and heart cutting function (AECS-QuikPrep™ Ltd, Cornwall, England). For cooling a circulation cooler is used (MC 600 Microcool, Lauda, Lauda-Königshofen, Germany).
- Preparative high pressure liquid chromatography (HPLC) with UV detector for four different wavelengths between 190-800 nm (TOY20DAD 800 H, Ecom, Chrastany u Prahy, Czech Republic ), Pump with a Flow of 0.001 to 50 mL/min (ECP2050, Ecom), four-way gradient valve (ECB2007, Ecom), fraction collector (ECF2096, Ecom), 2-channel online degasser (A5328, Knauer, Berlin, Germany) and various injection loops (2 mL, 500 μL and 20 μL). The system has the possibility of flash chromatography.
- Preparative purification by HPLC
- Semi- and preparative HPLC: ESI-LC-MS
-
Spectroscopy
- Spectrophotometry
- Electron spin resonance spectroscopy (ESR)
- Circular dichroism spectroscopy
- 1D and 2D NMR spectroscopy (Bruker Ascend 600 MHz with Avance NEO console, sample case and cryo sample head DUL)
-
Photometric assay
Photometric assays to determine the radical scavenging and total phenolic activities (TEAC, ORAC, FRAP, CUPRAC and DPPH).
Cell culture and molecular biology
-
Cultivation of various cell lines in the S I laboratory
e.g. Liver (HepG2, Huh7), macrophages (RAW246.7), intestines (Caco-2, HT-29), ovaries (CHO).
-
Toxicity assays
- Cytotoxicity assays (neutral red, LDH, MTT, WST-1, SRB, Resazurin, ATP, plating efficiency)
- Genotoxicity assays (Comet, Micronucleus)
- Mutagenicity assays (H(P)GRT, Ames)
-
Molecular biology
- Reporter gene assays
- Transfection
- Primer design
- Real-time PCR (QuantStudio™ 3)
- Microvolume UV/VIS spectrophotometer (NanoDrop One C; 190-850 nm)
- Western blot imaging system (iBright™ FL1500)
- ELISA (Tecan Infinite M200 with two injectors, absorbance (230-1000 nm), fluorescence (230 to 600 nm), and chemiluminescence)
- Flow cytometry
-
Enzyme assays and bioavailability
- Enzyme assays (such as α-amylase, α-glucosidase)
- Bioavailability (Transwell system with Caco-2 cells)
Contact


30167 Hannover










